Customers & Deployments
10min
Read
Pharma Industry: Full Traceability and Regulatory Compliance
Regulatory compliance in pharmaceutical environments depends on one critical factor: complete and reliable process traceability. LoconIQ replaces manual logging with real-time spatial ground truth—automatically tracking every movement, process step, and interaction to ensure maximum quality, auditability, and compliance without introducing human error.

Daniel Neuhold
CEO

Table of contents
Share
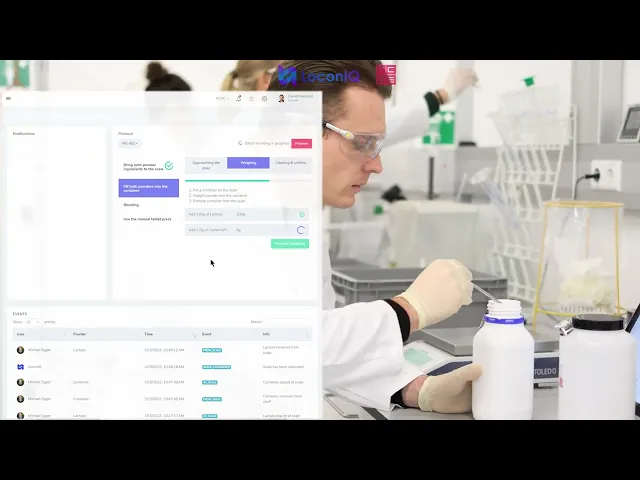
Eliminating Human Error in Pharmaceutical Operations
In pharmaceutical manufacturing and laboratory environments, even the smallest undocumented process deviation can create major compliance risks. Manual logging, handwritten records, barcode scans, and fragmented tracking systems introduce inefficiencies and leave room for human error.
LoconIQ transforms this process entirely.
By providing a real-time spatial ground-truth layer, LoconIQ automatically tracks assets, personnel, workflows, and process interactions with centimeter-level precision. Every movement, every transition between zones, and every process step is logged automatically—without requiring operators to manually enter data or interrupt workflows.
The result is a fully traceable and continuously validated operational environment.
Automated Process Documentation in Real Time
LoconIQ continuously monitors and documents operational workflows across pharmaceutical facilities, laboratories, and production environments.
This includes:
Movement tracking of sensitive assets and materials
Validation of process sequences and handling procedures
Automated timestamping of operational events
Monitoring of personnel interactions and zone access
Continuous audit trail generation for regulatory reporting
Verification that required process steps were completed correctly
Instead of relying on manual entries after the fact, the entire workflow is captured automatically in real time.
This significantly reduces administrative overhead while increasing data integrity and operational transparency.
Regulatory Compliance Built Into Operations
Compliance is no longer treated as a separate documentation task—it becomes part of the operational infrastructure itself.
LoconIQ enables pharmaceutical organizations to strengthen compliance with strict regulatory requirements by creating a deterministic and verifiable record of operational activities. Every action can be validated through precise spatial and temporal tracking.
The platform helps organizations:
Reduce compliance risks caused by incomplete documentation
Minimize human error in regulated workflows
Simplify audits through instantly accessible process histories
Increase operational quality assurance
Ensure full traceability across sensitive production environments
Because the platform operates fully on-premises, organizations maintain complete control over their operational and compliance data while benefiting from ultra-low latency and maximum data integrity.
A Smarter Foundation for Pharmaceutical Quality Assurance
Modern pharmaceutical environments require more than passive monitoring. They require intelligent infrastructure capable of validating operations continuously and reliably.
LoconIQ introduces a new approach where spatial intelligence becomes a core component of quality assurance and regulatory compliance. By automatically documenting and validating operational workflows in real time, pharmaceutical companies can improve reliability, reduce operational complexity, and ensure the highest standards of compliance without slowing down operations.



